DPP-4 Medication Safety Checklist
Step 1: Background Risk Factors
Select All That ApplyDo any of the following apply to your medical history?
Step 2: Watch List (Active Symptoms)
If you experience persistent symptoms, seek medical attention immediately.
-
CRITICAL
Severe stomach pain (often boring/deep)
May travel through to your back -
SYMPTOM
Nausea and Vomiting
Often accompany the abdominal pain
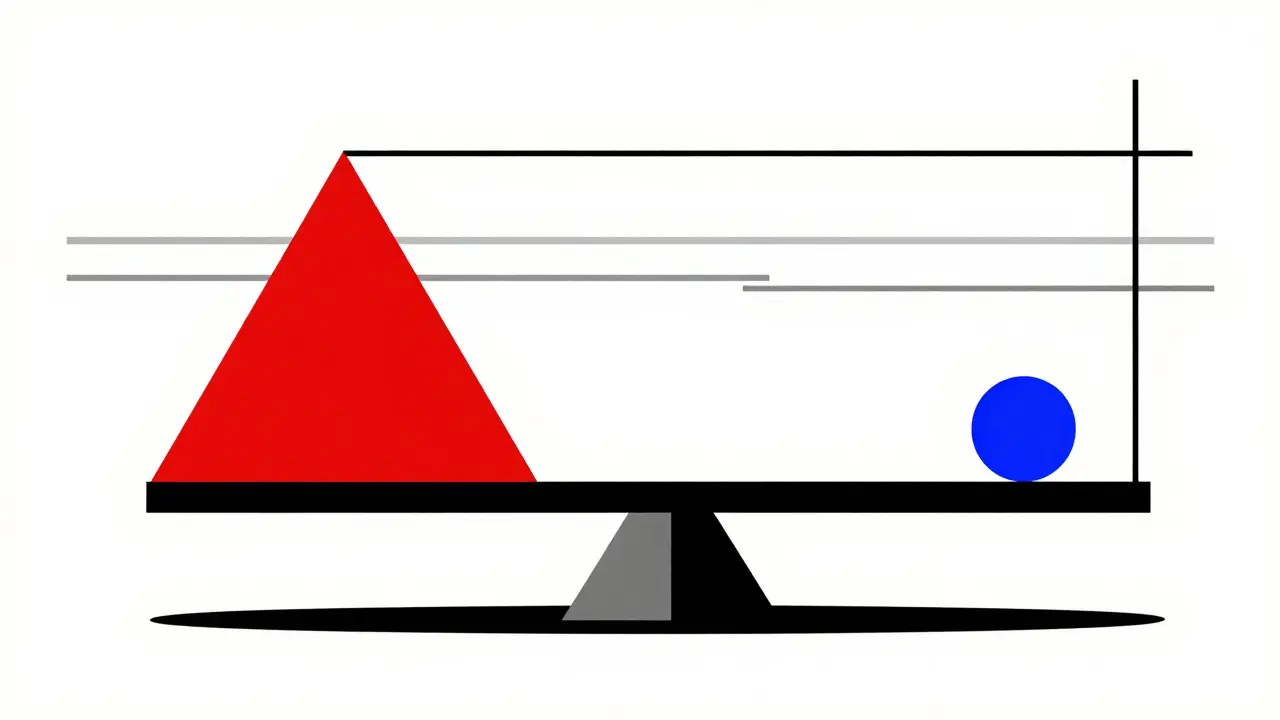
Understanding the Numbers
While relative risk increases are discussed, the absolute risk remains low for the majority.
1,000
Patients Treated Over 2 Years
~1 to 2
Additional Cases of Pancreatitis
0.13%
Estimated Absolute Increase
Based on pooled meta-analysis data including 47,714 patients.
The Reality of DPP-4 Inhibitors and Pancreatitis
If you have type 2 diabetes, you've likely heard of DPP-4 inhibitors. They are often prescribed because they help control blood sugar without causing weight gain or low blood sugar events. However, there is a persistent shadow over this class of medication. The question isn't whether they work, but rather, how safe are they regarding the pancreas? Specifically, does taking these pills put you at risk for acute pancreatitis?
This concern isn't just theoretical. Regulatory agencies worldwide have flagged it. Yet, the actual numbers tell a nuanced story that many patients miss. Understanding the difference between relative risk and absolute risk is crucial here. While studies show a statistical link, the real-world chance of developing this condition remains quite small for most people. Still, ignoring the warning signs is never an option.
- Quick Takeaways
-
- DPP-4 inhibitors carry a small but confirmed risk of acute pancreatitis.
- Absolute risk is approximately 0.13% extra over two years of treatment.
- Symptoms include severe abdominal pain that may travel to the back.
- Alternatives like SGLT2 inhibitors may have a lower risk profile.
- Patient awareness and early testing are vital for management.
Understanding DPP-4 Inhibitors
Before we discuss the risks, let's clarify exactly what we are talking about. DPP-4 Inhibitors, also known as "gliptins," are oral medications used to manage blood glucose levels in adults with type 2 diabetes. They work by blocking an enzyme called dipeptidyl peptidase-4. When you block this enzyme, your body keeps higher levels of incretin hormones. These hormones signal your pancreas to release insulin when you eat and stop releasing glucagon, a hormone that raises blood sugar.
Since their approval starting in 2006, several drugs have become staples in diabetes care. You might know them by their brand names: Januvia (sitagliptin), Onglyza (saxagliptin), Tradjenta (linagliptin), and Nesina or Vipidia (alogliptin). Each works similarly, but pharmacokinetics vary slightly. Linagliptin, for instance, is unique because it relies less on kidney function for clearance, making it a choice for patients with reduced kidney health. Despite their benefits, the association with pancreatic issues led to mandatory safety labeling updates by the Food and Drug Administration (FDA) in 2013.
The Statistics Behind the Warning
Data drives these safety warnings, but raw numbers can be confusing without context. Large-scale cardiovascular outcome trials involving tens of thousands of patients consistently showed a numerical imbalance. More cases of pancreatitis occurred in groups taking DPP-4 inhibitors compared to placebo groups. However, due to the rarity of the event, individual trials sometimes failed to reach statistical significance on their own. It was when data was pooled together that the pattern became undeniable.
One comprehensive meta-analysis involving 47,714 patients found that DPP-4 inhibitor treatment increased the risk of acute pancreatitis by roughly 75%. Another study cited in Frontiers in Pharmacology from 2024 reported a reporting odds ratio of 13.2. That sounds alarming until you look at the absolute risk. The same analysis noted the absolute increased risk is about 0.13%. To put this in plain English, treating 1,000 patients for two years might result in one to two additional cases of pancreatitis compared to a placebo group. It is a non-zero risk, but it affects a very small minority of users.
Regulatory bodies reflect this consensus. The UK Medicines and Healthcare products Regulatory Agency (MHRA) confirmed in September 2012 that while the risk was identified, the precise frequency remained difficult to pin down because pancreatitis is also more common in diabetics than in non-diabetic populations generally. This complicates the picture-is the drug causing it, or is it the underlying disease?
| Medication | Generic Name | Clinical Trial Findings |
|---|---|---|
| Januvia | Sitagliptin | First approved; numerical imbalance noted in CVOTs |
| Onglyza | Saxagliptin | Included in FDA meta-analysis; consistent signal |
| Tradjenta | Linagliptin | Small increase detected vs. placebo during development |
| Nesina/Vipidia | Alogliptin | Risk observed in large pooled datasets |
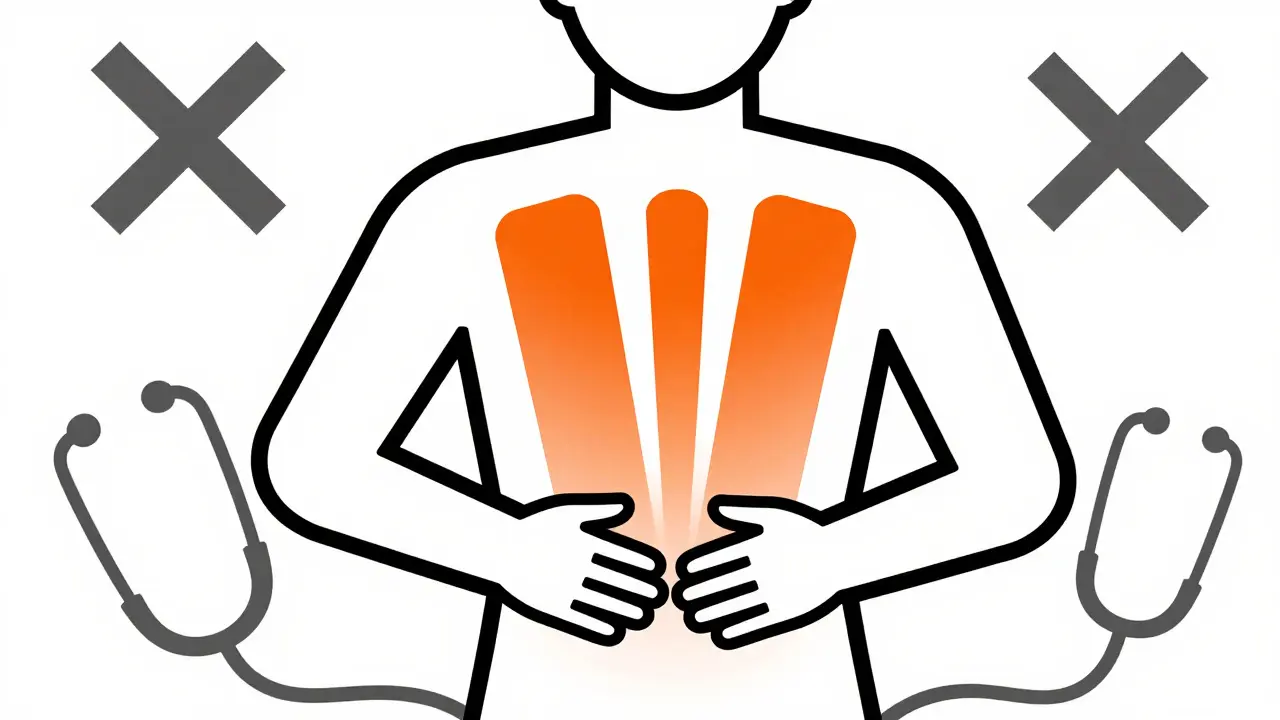
Recognizing the Symptoms Early
The most critical part of managing this risk is recognition. Acute pancreatitis is painful and serious, but catching it early prevents complications like chronic damage or infection. The hallmark symptom is persistent, severe abdominal pain. Often, patients describe this pain as boring deep into the abdomen and sometimes radiating to the back. Nausea and vomiting frequently accompany this pain.
If you are on a gliptin and feel this pain, do not wait. A 2017 guidance paper in Diabetes Care explicitly recommends measuring pancreatic enzymes (amylase and lipase) and performing an abdominal ultrasound immediately if such symptoms arise. The ultrasound helps rule out gallstones, which are another common cause of pancreatitis and can coexist with diabetes. Most cases resolve after discontinuing the drug, though about 17.7% of reported events involve serious outcomes according to 2024 data. Monitoring your liver function tests is also standard practice during therapy.
How They Compare to Other Options
When choosing a treatment path, knowing how these drugs compare to newer options is helpful. We have seen a shift toward SGLT2 inhibitors and GLP-1 receptor agonists due to heart and kidney benefits. But how do they stack up regarding pancreas safety?
Research indicates SGLT2 inhibitors generally have a significantly lower rate of acute pancreatitis compared to DPP-4 inhibitors. However, GLP-1 receptor agonists present their own considerations. Some studies suggest GLP-1s also associate with pancreatitis, with a reporting odds ratio of roughly 9.65, which is slightly lower than the 13.2 seen for DPP-4 inhibitors. Liraglutide, specifically, has been linked to this issue. Interestingly, despite fears about cancer, a major meta-analysis of 55,921 patients found neither drug class poses a significant risk for pancreatic cancer.
This comparison matters if you have a history of pancreatic inflammation, gallstones, or high alcohol consumption. In those high-risk scenarios, a clinician might lean toward a different class entirely to eliminate the variable. Conversely, if you have no history of pancreatic issues and require a medication that won't drop you into hypoglycemia, the gliptins remain a solid, evidence-backed choice.
Practical Steps for Patients
You might be reading this and wondering if you should stop your current medication today. Unless you have active symptoms, the answer is usually no. The American Diabetes Association continues to recommend DPP-4 inhibitors in its 2023 Standards of Medical Care. The benefit of controlling blood sugar to prevent long-term complications like nerve damage or blindness often outweighs the small risk of pancreatitis for most people.
However, being informed empowers you. Before starting treatment, ask your doctor about your personal risk factors. Have you ever had pancreatitis? Do you have a family history of it? Do you drink alcohol regularly? These factors compound the risk. Once on the medication, keep a close eye on your digestion. If discomfort arises, document it. Reporting adverse events through systems like the UK Yellow Card scheme or the FDA's Adverse Event Reporting System contributes to ongoing safety monitoring.
Living Safely with Your Treatment
Ultimately, the relationship between DPP-4 inhibitors and pancreatitis is managed, not eliminated. Pharmacovigilance programs like the FDA Sentinel Initiative continue to watch for trends. Real-world data from 1.2 million patients published in 2023 confirms the risk persists but stays low. Genetic markers are also under investigation to identify who specifically might react poorly to these drugs.
While global sales reached $5.8 billion in 2022, the conversation around safety ensures these medicines stay relevant. The future includes personalized medicine approaches, but for now, vigilance is the best policy. Keep communication lines open with your healthcare provider, report any stomach distress immediately, and trust in the monitoring systems that exist to protect you.
Is the risk of pancreatitis high with DPP-4 inhibitors?
No, the absolute risk is considered low. Studies estimate about 0.13% additional risk, meaning one to two extra cases per 1,000 patients treated over two years.
Can I continue taking DPP-4 inhibitors if I have diabetes?
Yes, for most patients, the benefits of blood sugar control outweigh the rare risk, provided you monitor for symptoms.
What are the main symptoms to watch for?
Look for persistent, severe abdominal pain that may radiate to the back, often accompanied by nausea or vomiting.
Are GLP-1 agonisms safer regarding pancreatitis?
They have a slightly lower risk profile in some studies (ROR 9.65 vs 13.2), but the risk is still present.
Do I need special blood tests to monitor this?
Routine monitoring isn't always required if asymptomatic, but if symptoms occur, pancreatic enzymes and ultrasounds are necessary.







Debra Brigman
March 29, 2026 AT 03:55The visceral nature of pancreatic vulnerability remains a haunting specter over our glucose management strategies. While we celebrate metabolic stability, the shadow of acute inflammation looms in the periphery of clinical data. We must acknowledge that biological systems possess an intricate fragility often masked by therapeutic gains. Each pharmaceutical intervention carries a latent potential for harm that persists despite rigorous monitoring protocols. Our collective health journey demands an honest reckoning with these subtle but real dangers.
tyler lamarre
March 29, 2026 AT 22:26Oh how delightful to witness such poetic drivel from the uneducated masses who lack proper scientific grounding. One presumes you believe emotional resonance substitutes for hard data derived from controlled trials. Your metaphorical prose ignores the fundamental statistical significance found within peer-reviewed literature. It is amusing to observe individuals prioritizing artistic flair over clinical precision in medical discussions.
Tony Yorke
March 31, 2026 AT 14:33stay focused on the facts here guys dont ignore the warnings keep watching your body signs if something feels wrong report it right away safety first always
Rachael Hammond
April 1, 2026 AT 11:49i think u r right about staying watchfull its scarry to hear the risk is real but low still we shud trust doctors more then fear maybe teh data is confusing
Devon Riley
April 1, 2026 AT 13:50Please remember that many patients manage these medications safely without any issues 🙌 Stay positive and communicate openly with your healthcare team 💊 Small risks exist but hope is powerful too 😊 Keep checking symptoms regularly ❤️
Tommy Nguyen
April 3, 2026 AT 10:30you got this everyone stay strong and informed talk to your doctor if needed dont panic about statistics focus on living well
Kameron Hacker
April 4, 2026 AT 18:44The implications of pharmacological negligence cannot be overstated in modern medical discourse. Patients are frequently exposed to systemic risks without adequate personal agency during prescription cycles. Mandatory reporting protocols must be enforced with greater scrutiny to protect public health integrity. Regulatory oversight fails when absolute risk is dismissed in favor of relative safety narratives. Vigilance remains the only defense against preventable adverse events.
Poppy Jackson
April 6, 2026 AT 06:13absolutely devastating thought that lives could hang in balance over enzyme readings truly shocking how little attention this gets deserves massive outcry
Jeannette Kwiatkowski Kwiatkowski
April 6, 2026 AT 14:02The statistical probability is negligible for the average patient profile regardless of marketing hype.
Aaron Olney
April 7, 2026 AT 14:12I readed the whole thing and got scared about what might happen next. My uncle had diabetes too and he suffered badly with his stomach. He always talked about his pain being unbearable sometimes. It was so bad you could see him sweat through his clothes. We didnt know what medicine caused it back then for sure. Maybe it was the gliptins we never understood fully back then. Now we have these fancy new stats everywhere showing small numbers. But numbers dont heal your belly pain when it burns inside. I feel worried when i take my morning pill without testing blood. Sometimes i wonder if it is safe really for someone like me. Doctors say its fine but my gut says no clearly to them. They want us to keep taking the meds anyway to control sugar. Risk is low but zero chance is better than any percent. I cannot understand why companies sell drugs that hurt people so much. Someone needs to fix this mess soon please help us out.